|
The relationship between mass, m, velocity, v, and wavelength,
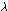 ,
is known as the de Broglie relationship, after the man who first
proposed it on theoretical grounds three years before the Davisson-Germer
experiment. ,
is known as the de Broglie relationship, after the man who first
proposed it on theoretical grounds three years before the Davisson-Germer
experiment.
This relationship is 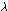 =
h/mv in which h is Planck's constant, the same constant we
encountered previously in the photoelectric effect. Since both mass
and speed are in the denominator of the expression, we can see that
a heavy particle or a rapidly moving one will have a short wavelength. =
h/mv in which h is Planck's constant, the same constant we
encountered previously in the photoelectric effect. Since both mass
and speed are in the denominator of the expression, we can see that
a heavy particle or a rapidly moving one will have a short wavelength.
The de Broglie wavelength relationship does not apply only to electrons.
It is valid for every particle in the universe, but the only particles
that have small enough mass that their wavelengths are long enough
to detect are electrons, protons, and neutrons.
In addition to x-ray and electron diffraction, neutron diffraction
is one of the standard tools of molecular structure analysis today.
|
|