|
Experimentally useful electron and neutron beams have wavelengths
from 1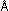 to a few hundredths of an angstrom. In contrast, a baseball thrown
at 30 meters per second has a de Broglie wavelength of only 10
to a few hundredths of an angstrom. In contrast, a baseball thrown
at 30 meters per second has a de Broglie wavelength of only 10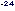
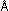 . .
This is less than one billion-billionth the diameter of an atomic
nucleus, and no experiment that we could ever design would reveal
the wave properties of a stream of baseballs. Wave properties of
particles only become apparent at the subatomic level.
The principle remains, however: All things in this universe are
both waves and particles, and have both a wavelength and an energy
per particle. At the size level encountered in everyday life, either
one or the other behavior predominates, which is why the wave-particle
duality was overlooked until this century. But at the atomic and
subatomic level, both wave and particle behavior can be detected
under the right experimental conditions. The dualism is real.
Electrons do not just assume wavelike behavior now and again like
a disguise; electrons are waves as well as particles. This is important
in the first modern theory of atomic structure, the Bohr picture
of the hydrogen atom.
|
|