|
Description
Ice cubes float on cooking oil but, on melting, the water that
is produced sinks.
Apparatus
One 1dm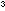 measuring cylinder.
measuring cylinder.
Chemicals
The quantities given are for one demonstration.
Water.
A few ice cubes. These may be made with a little food colouring
(blue is good) for better visibility.
400 cm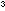 of cooking oil.
of cooking oil.
|
|
Method
Make the ice cubes with a few drops of food colouring per cube.
Ensure that they are completely frozen. Partly frozen cubes may
have liquid water trapped inside which will affect their density.
Check that ice cubes do actually float on the brand of cooking oil
to be used.
The Demonstration
Place about 400cm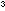 of water and 400cm
of water and 400cm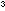 of cooking oil in the measuring cylinder. Allow the two layers to
separate fully; the oil will be on the top. Drop an ice cube into
the cylinder. It will float (just) on top of the oil. Watch the
cube. As it melts, the water that is formed makes a droplet attached
to the cube. Eventually this detaches itself from the cube and sinks,
joining the water layer below. This illustrates the anomalously
greater density of water compared to ice.
of cooking oil in the measuring cylinder. Allow the two layers to
separate fully; the oil will be on the top. Drop an ice cube into
the cylinder. It will float (just) on top of the oil. Watch the
cube. As it melts, the water that is formed makes a droplet attached
to the cube. Eventually this detaches itself from the cube and sinks,
joining the water layer below. This illustrates the anomalously
greater density of water compared to ice.
|