|
Theory
Glucose is a reducing agent and in alkaline solution will reduce
methylene blue to a colourless form. Shaking the solution admits
oxygen which will re-oxidise the methylene blue back to the blue
form.
Further details
A.G. Cook, R.M. tolliver and J.E. Williams, J. Chem. Ed.,
1994, 71, 160. The article The blue bottle experiment
revisited gives some details of the reaction mechanism and alternate
dyes.
Safety
Wear eye protection.
|
|
Further Tips
A white laboratory coat provides an ideal background. On a cold
day, it may be necessary to warm the solution to 25-30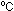 or the colour changes will be very slow. The demonstration can be
used to start a discussion on what is causing the colour changes.
Students' suggestions can be tried out as far as practicable.
or the colour changes will be very slow. The demonstration can be
used to start a discussion on what is causing the colour changes.
Students' suggestions can be tried out as far as practicable.
Extensions
To confirm that oxygen is responsible for the colour change,
nitrogen can be bubbled through the solution for a couple of minutes
to displace the air from the solution and the flask. If the stopper
is now replaced and the bottle shaken, no colour change will occur.
Reintroducing the air by pouring the solution into another flask
and shaking will restore the system.
|