Boron-Oxygen Compounds
2B + 3/2O2 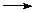 B2O3
B2O3 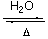 B(OH)3
Boric oxide Boric acid
Structure-H-bonded: planar B(O)3
unit p-bonding
Acidic => generates protons
B(OH)3 + 2H2O
B(OH)3
Boric oxide Boric acid
Structure-H-bonded: planar B(O)3
unit p-bonding
Acidic => generates protons
B(OH)3 + 2H2O 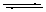 B(OH)4- + H3O+
cf. HCl + H2O
B(OH)4- + H3O+
cf. HCl + H2O 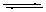 Cl- + H3O+
NB .
Cl- + H3O+
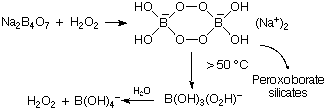
NB . 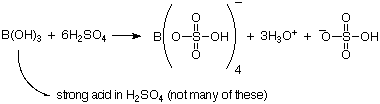 B2O3 + metal oxide
B2O3 + metal oxide  Borates (Again acid properties)
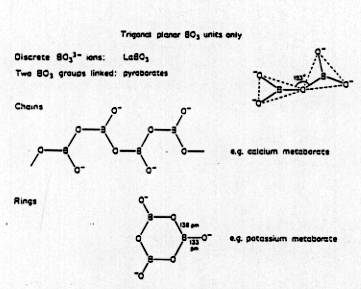
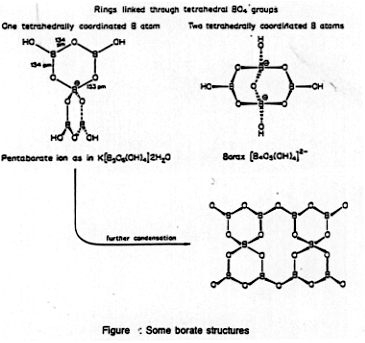
Structures
- BO3 and tetrahedral BO4
units, no BO6 units (due to size)
- BO33- + H2O
Borates (Again acid properties)
Structures
- BO3 and tetrahedral BO4
units, no BO6 units (due to size)
- BO33- + H2O  B-O-B linkages
B-O-B linkages
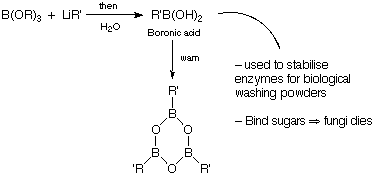
 B(OH)3 + ROH
B(OH)3 + ROH  B(OR)(OH)2
B(OR)(OH)2  B(OR)2OH,
B(OR)3
Borate esters
B(OR)2OH,
B(OR)3
Borate esters
 B2O3
B2O3  B(OH)3
B(OH)3 B2O3
B2O3 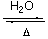 B(OH)3
B(OH)3 B(OH)4- + H3O+
B(OH)4- + H3O+ Cl- + H3O+
Cl- + H3O+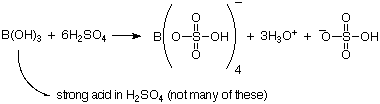
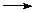 Borates (Again acid properties)
Borates (Again acid properties)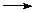 B-O-B linkages
B-O-B linkages
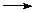 B(OR)(OH)2
B(OR)(OH)2 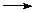 B(OR)2OH,
B(OR)3
B(OR)2OH,
B(OR)3

