|
Chemicals
About 1.5 dm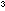 of liquid nitrogen will be enough to do all of the demonstrations
described.
of liquid nitrogen will be enough to do all of the demonstrations
described.
Lead sheeting, about 1-2 mm thick.
A few cm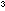 of mercury.
of mercury.
Access to an oxygen cylinder and regulator.
Access to a hydrogen cylinder and regulator.
Access to a carbon dioxide cylinder with regulator.
A little washing up liquid and some food dye (optional).
Bubble mixture.
One banana and/or flower.
Method
1. Pour some liquid nitrogen into an expanded polystyrene box
and immerse a variety of items into it.
(a) Mercury (in a small test-tube) solidifies.
|
|
(b) Rubber or plastic tubing goes brittle and can be snapped in
gloved hands or shattered with a sharp blow.
(c) A strip of lead becomes rigid and will 'ring' when tapped with
another metal.
(d) A banana, for example, will become brittle and will shatter
if it is hit on the bench. Take care to clear up all the shattered
pieces - they become very soggy on melting!
(e) Flowers or leaves will become brittle.
(f) A ballon blown up with air (by mouth) will shrink as the air
liquefies. Liquid air can be seen and felt inside the balloon. The
rubber attains a texture rather like a crisp packet. The change
is fully reversible. A balloon blown up from a hydrogen cylinder
shrinks to about 1/3 of its original size because in this case the
gas will not liquefy. The balloon expands back to its original size
when it is removed. If a suitably sized balloon is used, the balloon
will sink to the floow when shrunken and will float as it warms
up and expands. These are dramatic examples of the gas laws and
deviations from them.
(g) A soap bubble can be held on a wire 'wand' over a container
of liquid nitrogen. It will freeze.
|