|
In
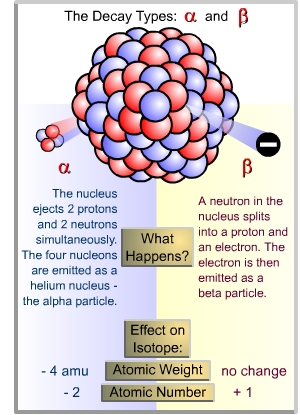
the transformations on the previous page, the greek symbol alpha
indicates alpha-particle emission, and beta represents beta
decay.
Excercise.
Write a few of the individual steps to check that mass number and
charge are conserved.
In this series of breakdowns, uranium emits an alpha particle to
become an unstable isotope of thorium, which goes via 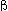 decay to protactinium and then to a different isotope of uranium.
A series of a decays then descends through thorium, radium, radon,
and polonium to an unstable lead isotope, and the process continues
until a stable form of lead is reached. The entire
decay to protactinium and then to a different isotope of uranium.
A series of a decays then descends through thorium, radium, radon,
and polonium to an unstable lead isotope, and the process continues
until a stable form of lead is reached. The entire  decay chain represented on a p-n stability plot can be viewed on
the previous page. Another decay chain leads from
decay chain represented on a p-n stability plot can be viewed on
the previous page. Another decay chain leads from 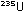 to
to  , and
a third chain, not shown, begins with , and
a third chain, not shown, begins with  and ends at
and ends at 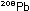 .
Lead is just at the upper end of the stable region, as seen on Page
29, and the heavier elements beyond it to the upper right decay
spontaneously back to lead. .
Lead is just at the upper end of the stable region, as seen on Page
29, and the heavier elements beyond it to the upper right decay
spontaneously back to lead.
|
|
